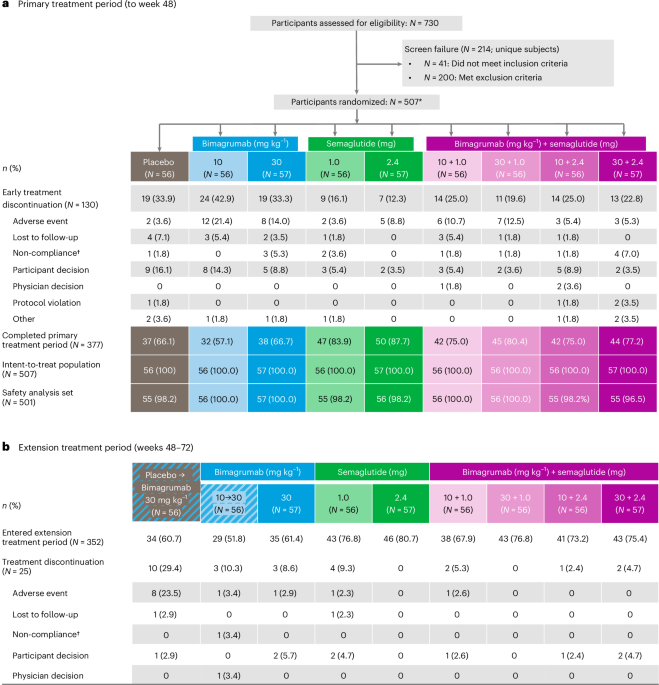
Patient disposition
From 16 November 2022 to 16 May 2024, 730 participants were screened for eligibility. Of these, 507 were randomized to placebo (n = 56), bimagrumab (10 mg kg−1: n = 56; 30 mg kg−1: n = 57), semaglutide (1.0 mg: n = 56; 2.4 mg: n = 57) and bimagrumab plus semaglutide (combination; 10 mg kg−1 plus 1.0 mg: n = 56; 30 mg kg−1 plus 1.0 mg: n = 56; 10 mg kg−1 plus 2.4 mg: n = 56; 30 mg kg−1 plus 2.4 mg: n = 57) groups (Fig. 1). Overall, 377 (74.4%) participants completed the primary treatment period at week 48. Treatment discontinuations due to adverse events were higher in the bimagrumab groups (14.0–21.4%) than in the semaglutide (3.6–8.8%), combination (5.3–12.5%) and placebo (3.6%) groups. During the extension period, 25 (7.1%) participants discontinued treatment (Fig. 1).
*12 participants met eligibility criteria but were not randomized, and three participants did not meet all eligibility criteria but were randomized. †Non-compliance with study requirements. a, Participant disposition during primary treatment period (to week 48). b, Participant disposition during extension treatment period (weeks 48 to 72).
Baseline demographics and clinical characteristics
Demographic and clinical baseline characteristics were largely similar across treatment groups; most participants were female (57.4%) and White (75.1%) (Table 1). Mean values for the trial were as follows: age 47.5 years, body weight 107.5 kg, body mass index (BMI) 37.3 kg m−2, waist circumference 118.1 cm, total body fat mass (by dual-energy X-ray absorptiometry (DXA)) 45.8 kg and total body lean mass (by DXA) 58.3 kg (Table 1).
Primary outcome
For the efficacy results at week 48, nominal P values versus placebo and versus semaglutide 2.4 mg are reported in Table 2 and Fig. 2.
*P < 0.05, **P < 0.01, ***P< 0.001 active treatment groups versus placebo. #P < 0.05, ##P < 0.01, ###P < 0.001 active treatment groups versus semaglutide 2.4 mg. Data are presented as LSM change from baseline ± standard error. P values for comparisons with placebo and semaglutide 2.4 mg were calculated using two-sided t-tests without multiplicity adjustment. n values are provided in the source data tables in the supplementary information. a−e, The LSM percent or absolute changes from baseline to week 48 in efficacy endpoints are based on an MMRM analysis for the efficacy estimand and an ANCOVA model with multiple imputation for the treatment regimen estimand. f, The LSM percent changes in hsCRP from baseline to week 48 are based on MMRM analysis using log transformation for the efficacy estimand. ANCOVA, analysis of covariance; hsCRP, High-sensitivity C-Reactive Protein; LSM, least-squares mean; MMRM, mixed model for repeated measures; VAT, visceral adipose tissue.
Source data
Absolute change in body weight
Treatment regimen estimand
At week 48, the least squares mean (LSM) absolute change in body weight was −6.0 kg to −9.3 kg (bimagrumab), −9.8 kg to −14.2 kg (semaglutide) and −12.7 kg to −17.8 kg (combination) versus −3.3 kg (placebo) (all P < 0.001 versus placebo, except bimagrumab 10 mg kg−1; Table 2). The LSM change in absolute body weight was greater with the high-dose combination versus semaglutide 2.4 mg (−17.8 kg versus −14.2 kg; P < 0.05).
Secondary outcomes
Percent change in body weight
Treatment regimen estimand
The LSM percent change in body weight was −5.6% to −8.6% (bimagrumab), −9.8% to −13.5% (semaglutide) and −12.0% to −16.4% (combination) compared to −3.5% (placebo) (Fig. 2a). By week 48, ≥15% weight reduction was achieved in 23.3% (bimagrumab 30 mg kg−1), 43.4% (semaglutide 2.4 mg) and 63.9% (high-dose combination) of participants (Extended Data Fig. 2).
Efficacy estimand
The LSM percent change in body weight at week 48 was −5.0% to −9.7% (bimagrumab), −11.0% to −14.8% (semaglutide) and −14.3% to −20.2% (combination) versus −2.5% (placebo) (P < 0.001 for the high-dose combination versus semaglutide 2.4 mg; Fig. 2a). At week 72, the LSM change in weight was −12.0 kg (−10.8%; bimagrumab 30 mg kg−1), −16.5 kg (−15.7%; semaglutide 2.4 mg) and −24.2 kg (−22.1%; high-dose combination) (Fig. 3a and Extended Data Table 1). By week 72, ≥15% weight reduction was achieved in 21.8% (bimagrumab 30 mg kg−1), 51.8% (semaglutide 2.4 mg) and 84.9% (high-dose combination) of participants (Extended Data Fig. 2).
Data are presented as LSM change from baseline ± standard error. n represents the number of participants with baseline and post-baseline values at week 72. a−e, The LSM percent or absolute changes from baseline at week 72 in efficacy endpoints are based on MMRM analysis for the efficacy estimand. f, The LSM percent changes in hsCRP from baseline to week 72 are based on MMRM analysis using log transformation for the efficacy estimand. hsCRP, high-sensitivity C-reactive protein; LSM, least-squares mean; MMRM, mixed model for repeated measures; VAT, visceral adipose tissue.
Source data
Waist circumference
Treatment regimen estimand
Absolute LSM change in waist circumference at week 48 was −7.3 cm to −9.1 cm (bimagrumab), −8.7 cm to −12.1 cm (semaglutide) and −12.4 cm to −15.8 cm (combination) compared to −4.6 cm (placebo) (Fig. 2b). Results for improvements in waist-to-height ratio categories at week 48 are provided in Extended Data Fig. 3.
Efficacy estimand
Absolute LSM changes in waist circumference at week 48 were −8.1 cm to −10.7 cm (bimagrumab), −10.5 cm to −13.6 cm (semaglutide) and −14.7 cm to −19.4 cm (combination) compared to −4.7 cm (placebo) (P < 0.001 for the high-dose combination versus semaglutide 2.4 mg; Fig. 2b). At week 72, absolute LSM changes in waist circumference were −13.3 cm (bimagrumab 30 mg kg−1), −14.7 cm (semaglutide 2.4 mg) and −21.7 cm (high-dose combination) (Fig. 3b).
Body composition (by DXA)
Treatment regimen estimand
The LSM percent reduction in total body fat mass at week 48 was −13.0% to −18.9% (bimagrumab), −16.0% to −21.1% (semaglutide) and −24.7% to −33.7% (combination) compared to −5.6% (placebo) (P < 0.001 for the high-dose combination versus semaglutide 2.4 mg; Fig. 2c). Mean percent body fat decreased from 43.7% at baseline to 32.5% at week 48 with high-dose combination compared to 41.9% to 37.9% with semaglutide 2.4 mg. By week 48, fat mass reduction ≥25% was achieved in 30.8% (bimagrumab 30 mg kg−1), 36.3% (semaglutide 2.4 mg) and 73.6% (high-dose combination) of participants (Extended Data Fig. 4).
The LSM percent changes in total body lean mass at week 48 were +1.0% to +1.1% (bimagrumab), −4.7% to −6.9% (semaglutide) and −0.8% to −2.3% (combination) versus −0.9% (placebo) (P < 0.001 for all combination groups versus semaglutide 2.4 mg; Fig. 2d). At week 48, the proportion of weight loss due to fat mass (fat loss index) was 100% (bimagrumab 30 mg kg−1), 71.1% (semaglutide 2.4 mg) and 92.3% (high-dose combination). The bimagrumab 30 mg kg−1 plus semaglutide 1.0 mg group showed the greatest preservation of lean mass among the combination groups. Results for appendicular lean mass at week 48 are presented in Table 2.
The percent LSM changes in estimated VAT were −15.7% to −26.7% (bimagrumab), −19.3% to −24.5% (semaglutide) and −33.1% to −43.8% (combination) versus −3.3% (placebo) (P < 0.01 for the high-dose combination versus semaglutide 2.4 mg; Fig. 2e).
Efficacy estimand
The LSM percent reduction in total body fat mass at week 48 was −15.9% to −25.3% (bimagrumab), −18.4% to −24.8% (semaglutide) and −30.6% to −42.2% (combination) compared to −5.2% (placebo) (P < 0.05 for all combination groups versus semaglutide 2.4 mg; Fig. 2c). At week 72, LSM percent reductions in total body fat mass were −28.5% (bimagrumab 30 mg kg−1), −27.8% (semaglutide 2.4 mg) and −45.7% (high-dose combination) (Fig. 3c). By week 72, fat mass reduction ≥30% was achieved in 50.0% (bimagrumab 30 mg kg−1), 36.4% (semaglutide 2.4 mg) and 94.0% (high-dose combination) of participants (Extended Data Fig. 4).
The LSM percent changes in total body lean mass at week 48 were +2.3% to +2.7% (bimagrumab), −5.3% to −7.9% (semaglutide) and −1.1% to −2.6% (combination) versus −0.5% (placebo) (P < 0.001 for all combination groups versus semaglutide 2.4 mg; Fig. 2d). At week 72, LSM changes in total body lean mass were +2.5% (bimagrumab 30 mg kg−1), −7.4% (semaglutide 2.4 mg) and −2.9% (high-dose combination) (Fig. 3d). Results for appendicular lean mass at week 72 are presented in Extended Data Table 1.
At week 72, the proportion of weight loss due to fat mass was 100% (bimagrumab 30 mg kg−1), 75.6% (semaglutide 2.4 mg) and 92.2% (high-dose combination) (Extended Data Table 1).
At week 48, the percent LSM changes in estimated VAT were −23.0% to −40.2% (bimagrumab), −21.5% to −29.5% (semaglutide) and −41.0% to −54.8% (combination) versus −2.1% (placebo) (P < 0.01 for all combination groups versus semaglutide 2.4 mg; Fig. 2e). At week 72, reductions in estimated VAT were −45.1% (bimagrumab 30 mg kg−1), −35.8% (semaglutide 2.4 mg) and −58.2% (high-dose combination) (Fig. 3e).
Metabolic parameters and physical function
HbA1c
HbA1c levels improved in semaglutide and combination groups at week 48 (Table 2; treatment regimen estimand). The LSM decreases in HbA1c levels at week 72 were −0.23% (bimagrumab 30 mg kg−1), −0.40% (semaglutide 2.4 mg) and −0.55% (high-dose combination) (Extended Data Table 1; efficacy estimand). Among participants with HbA1c ≥ 5.7% at baseline (prediabetes), normoglycemia (defined as HbA1c < 5.7%) at week 48 was achieved in 19 of 29 participants (66%, bimagrumab groups), in 23 of 27 participants (85%, semaglutide groups) and in 44 of 45 participants (98%, combination groups), compared to six of 15 participants (40%) in the placebo group (Table 2; efficacy estimand). At week 72, normoglycemia was achieved in 22 of 29 participants (76%, bimagrumab groups), in 26 of 27 participants (96%, semaglutide groups) and in 45 of 45 participants (100%, combination groups) compared to eight of 15 participants (53%) in the placebo group (Extended Data Table 1; efficacy estimand).
Patient-reported outcomes
Treatment regimen estimand
Improvements in 36-Item Short Form Health Survey (SF-36) Physical Functioning score were similar across treatment groups at week 48 (P value not significant versus placebo; Table 2). Combination groups containing bimagrumab 30 mg kg−1 showed greater improvements in Impact of Weight on Quality of Life-Lite Clinical Trials Version (IWQOL-Lite-CT) Physical Function scores at week 48 compared to the placebo group (P < 0.05; Table 2).
Efficacy estimand
At week 72, improvements in SF-36 Physical Functioning scores and IWQOL-Lite-CT Physical Function scores were greater in the high-dose combination group than in the remaining groups (Extended Data Table 1).
Safety
Overall, safety results during the primary treatment period were consistent with known safety profiles of the two drugs. The incidence of treatment-emergent adverse events during the primary treatment period was similar among active drug treatment groups (91.1−98.2%) and greater than placebo (74.5%) (Table 3). Serious adverse events were reported in 8.8−12.5% of participants (bimagrumab), in 1.8−10.7% of participants (semaglutide) and in 3.6−9.1% of participants (combination) versus in 3.6% of participants (placebo); no deaths were reported. Common adverse events included muscle spasms (commonly, muscle cramps), diarrhea and acne with bimagrumab and nausea, diarrhea, constipation and fatigue with semaglutide, with similar events in the combination groups. All treatment discontinuations due to nausea (N = 6) occurred in the combination groups, and treatment discontinuations due to muscle spasms (N = 5) occurred in the bimagrumab monotherapy groups (Table 3). Four discontinuations were due to acne: two in the bimagrumab monotherapy groups and two in the combination groups.
Adverse events of special interest
Primary treatment period (through week 48)
Thirteen participants had severe (grade 3) gastrointestinal-related events, including three with pancreatitis serious adverse events (one each in placebo, bimagrumab 10 mg kg−1 and semaglutide 1.0 mg groups). One participant had severe acne and five had severe muscle-related events (muscle spasms and back pain) in bimagrumab-containing groups (Table 3). Four participants reported basal or squamous cell skin carcinoma (all in bimagrumab-only or semaglutide-only groups); no other malignancies were reported (Table 3).
Open-label extension treatment period (weeks 48–72)
There were no new safety signals during weeks 48–72 (Extended Data Table 2). Two participants had severe gastrointestinal-related events (bimagrumab 10 mg kg−1 plus semaglutide 2.4 mg: abdominal pain; bimagrumab 30 mg kg−1 plus semaglutide 1.0 mg: enteritis); one participant had severe muscle-related events (back pain; placebo switched to bimagrumab 30 mg kg−1 group); and two participants reported basal cell skin carcinoma (bimagrumab 30 mg kg−1 and semaglutide 1.0 mg groups) (Extended Data Table 2).
Laboratory parameters
No clinically relevant changes in hematologic or renal parameters were observed. Mean magnesium levels decreased in bimagrumab-containing groups but remained within the normal range across treatment groups. Bimagrumab-containing groups showed mean increases in alkaline phosphatase (ALP) and creatine kinase, transient increases in alanine aminotransferase (ALT) (Extended Data Fig. 5) and aspartate aminotransferase (AST) and decreases in total bilirubin and gamma-glutamyl transferase (GGT). Serum lipase increased transiently with bimagrumab but increased and remained elevated with semaglutide treatment (Extended Data Fig. 5).
Blood pressure (efficacy estimand)
The mean decreases in systolic blood pressure (SBP) were not significantly different between treatment groups at week 48 (bimagrumab: −3.1 mmHg to −4.6 mmHg; semaglutide: −7.9 mmHg to −8.6 mmHg; combination: −4.5 mmHg to −10.4 mmHg; placebo: −6.9 mmHg) (Table 2). The mean reduction in diastolic blood pressure (DBP) was greater in the high-dose combination group versus the placebo and semaglutide 2.4 mg groups at week 48 (−6.7 mmHg versus −2.8 mmHg and −3.4 mmHg, respectively; P < 0.05) (Table 2).
Exploratory outcomes
Bone mineral density (efficacy estimand)
The LSM percent changes in total body and lumbar spine bone mineral density (BMD) were ≤1.1% in all groups at week 48 (Table 2). The LSM percent decreases in total hip BMD were significantly greater in the semaglutide 2.4 mg (−2.1%) group, the bimagrumab 10 mg kg−1 plus semaglutide 1 mg (−2.0%) group and the two combination groups containing bimagrumab 30 mg kg−1 (−2.2% to −2.3%) versus placebo (−0.8%) (P < 0.05). The LSM percent changes from baseline in femoral neck BMD were not significantly different in the treatment groups versus placebo (Table 2). Changes in these BMD outcomes at week 72 were similar, with greater decreases in total hip and/or femoral neck BMD than in total body or lumbar spine BMD across groups (Extended Data Table 1).
Lipids (efficacy estimand)
Total and low-density lipoprotein (LDL) cholesterol levels increased in the first 12 weeks in the bimagrumab-containing groups and then decreased toward baseline in the combination groups containing semaglutide 2.4 mg while remaining above baseline in the bimagrumab-only groups and the combination groups containing semaglutide 1.0 mg (Table 2, Extended Data Table 1 and Extended Data Fig. 6). By contrast, increases in high-density lipoprotein (HDL) cholesterol and decreases in triglyceride levels were similar in the combination and semaglutide-only groups at weeks 48 and 72 (Table 2, Extended Data Table 1 and Extended Data Fig. 6). At week 72, LSM percent changes in LDL cholesterol were 17.6% (bimagrumab 30 mg kg−1), −8.9% (semaglutide 2.4 mg) and 0.1% (high-dose combination) (Extended Data Fig. 6). For triglycerides, these were −1.2% (bimagrumab 30 mg kg−1), −20.8% (semaglutide 2.4 mg) and −25.3% (high-dose combination) (Extended Data Fig. 6).
High-sensitivity C-reactive protein (efficacy estimand)
The LSM percent reductions in high-sensitivity C-reactive protein (hsCRP) at week 48 were −52.9% to −69.0% (bimagrumab), −54.5% to −55.4% (semaglutide) and −71.5% to −83.1% (combination) versus −15.6% (placebo group) (Fig. 2f). At week 72, the LSM percent reductions in hsCRP were −72.0% (bimagrumab 30 mg kg−1), −59.3% (semaglutide 2.4 mg) and −84.0% (high-dose combination) (Fig. 3f).
Adipokines, fasting insulin and free testosterone (efficacy estimand)
The LSM changes in serum leptin levels at week 48 were −20.5 ng ml−1 to −26.6 ng ml−1 (bimagrumab), −20.1 ng ml−1 to −20.5 ng ml−1 (semaglutide) and −24.9 ng ml−1 to −31.9 ng ml−1 (combination) versus −7.7 ng ml−1 (placebo) (Extended Data Fig. 7). The LSM increases in serum adiponectin levels at week 48 were 14.1 µg ml−1 to 28.6 µg ml−1 (bimagrumab), 6.2 µg ml−1 to 7.5 µg ml−1 (semaglutide) and 22.0 µg ml−1 to 33.3 µg ml−1 (combination) versus 4.8 µg ml−1 (placebo) (Extended Data Fig. 7).
The LSM changes in fasting insulin levels at week 48 are presented in Table 2. At week 72, the LSM decreases in fasting insulin were greater in the high-dose combination group (−34.4 pmol l−1) versus bimagrumab 30 mg kg−1 (−26.3 pmol l−1) and semaglutide 2.4 mg (−27.6 pmol l−1) groups (Extended Data Table 1). The LSM changes in free testosterone levels are presented in Table 2 (week 48) and Extended Data Table 1 (week 72).
Grip strength (efficacy estimand)
The bimagrumab 30 mg kg−1 plus semaglutide 1.0 mg group showed the greatest increase in grip strength among treatment groups at week 48 (4.8 kg; P < 0.05 versus placebo (1.7 kg)) (Table 2); all remaining groups were similar to placebo. There was a trend for increased grip strength in the bimagrumab monotherapy groups that was not significantly different compared to semaglutide 2.4 mg at week 48. Results at week 72 are presented in Extended Data Table 1.
Dietary intake
At week 24, the median change in total calories (kcal d−1) in the higher dose groups was −182.0 (bimagrumab 30 mg kg−1), −482.5 (semaglutide 2.4 mg) and −487.0 (high-dose combination) versus −238.5 (placebo). At week 48, participants in the semaglutide 2.4 mg and placebo groups had a greater median reduction in total caloric intake than those in the other groups (Table 2). The median increase in protein intake (as % total calories) was highest in the bimagrumab and placebo groups compared to the semaglutide and combination groups (Table 2).