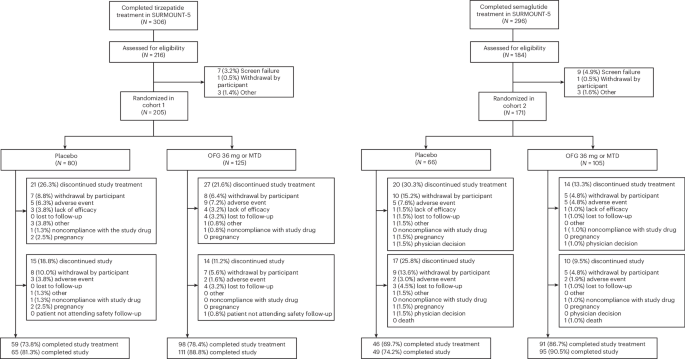
ATTAIN-MAINTAIN screened 400 and enrolled 376 participants between 13 September 2024 and 21 November 2025, in 29 sites in the USA. A total of 205 participants in cohort 1 were randomized to treatment (N = 125 orforglipron 36 mg or maximum tolerated dose (MTD), N = 80 placebo) and 171 participants in cohort 2 were randomized to treatment (N = 105 orforglipron 36 mg or MTD, N = 66 placebo) (Fig. 1). This work reports data on the investigational orforglipron capsule formulation of 1, 3, 6, 12, 24 and 36 mg; the doses have been shown as the equivalent to tablet doses of 0.8, 2.5, 5.5, 9, 14.5 and 17.2 mg, respectively, which are approved in the USA13. Overall, 157 (76.6%) participants in cohort 1 and 137 (80.1%) participants in cohort 2 completed study treatment, and 176 (85.9%) completed the study in cohort 1 and 144 (84.2%) in cohort 2. Reasons for early study treatment and study discontinuations are shown in Fig. 1. Table 1 presents the demographic and baseline characteristics of the participants. In cohort 1, the mean age of the participants was 48.5 years; most were female (62.9%) and white (73.8%) and had a mean body weight of 90.1 kg. In cohort 2, the mean age was 48.6 years; most participants were female (68.4%) and white (75.9%) and had a mean body weight of 94.4 kg.
CONSORT diagrams summarizing the design of the double-blind, placebo-controlled trial in which randomized participants previously treated with either tirzepatide (cohort 1: N = 205) or semaglutide (cohort 2: N = 171) during the SURMOUNT-5 study were treated with the GLP-1 receptor agonist orforglipron (OFG) once daily or with placebo.
Cohort 1: tirzepatide to orforglipron (MTD) versus placebo
For the primary endpoint with the modified treatment-regimen estimand, participants in cohort 1 who achieved body weight plateau (<5% body weight change from weeks 60 to 72 in the SURMOUNT-5 study) maintained 74.7% (s.e.m. 4.05) of body weight reduction with orforglipron compared with 49.2% (s.e.m. 3.92) with placebo, with an estimated treatment difference (ETD) relative to placebo of 25.5% (95% confidence interval (CI) 14.5 to 36.5); P < 0.001) at week 52. Key secondary endpoints in the modified treatment-regimen estimand, in cohort 1 participants who achieved a body weight plateau, 43.7% (s.e.m. 4.98) of participants treated with orforglipron maintained ≥80% of the body weight reduction achieved during SURMOUNT-5 compared with 16.4% (s.e.m. 4.60) with placebo, with a risk difference to placebo of 27.3 (95% CI 14.1 to 40.6); P < 0.001 (efficacy estimand data shown in Extended Data Fig. 1a). All participants in cohort 1 maintained 74.4% (s.e.m. 3.63) of body weight reduction with orforglipron and 49.7% (s.e.m. 3.53) with placebo, with an ETD relative to placebo of 24.7 percentage points (95% CI 14.9 to 34.5; P < 0.001) with orforglipron treatment. All participants in cohort 1 had a mean MBE percent change from SURMOUNT-5 baseline body weight of –16.5% (s.e.m. 0.90) with orforglipron and 12.6% (s.e.m. 0.91) with placebo with an ETD relative to placebo of –3.9 percentage points (95% CI –6.1 to –1.8; P < 0.001).
In additional sensitivity analyses based on the efficacy estimand, participants in cohort 1 randomized only to orforglipron in ATTAIN-MAINTAIN had a mean baseline body weight of 115.8 kg (s.e.m. 2.23) in SURMOUNT-5. At the start of the ATTAIN-MAINTAIN study, they had a baseline body weight of 90.9 kg (s.e.m. 2.12) with a reduction of 21.5% (s.e.m. 0.83). Participants in cohort 1 had a mean MBE body weight reduction of 16.8% (s.e.m. 0.76) (Fig. 2a) and absolute body weight reduction of 19.6 kg (s.e.m. 0.91) (Fig. 2b) from the beginning of SURMOUNT-5 to the end of ATTAIN-MAINTAIN. This represents a difference in percent weight changes of approximately 5% and an average difference in weight of approximately 5 kg from baseline. For these participants, the mean body weight (MBE) at week 52 was 95.9 kg (s.e.m. 0.91) with orforglipron (Fig. 2c). Participants in cohort 1 at 24 weeks, before rescue therapy eligibility per study protocol, had a mean MBE body weight reduction of −19.0% (s.e.m. 0.68) from the beginning of SURMOUNT-5, with a mean MBE body weight of 93.4 kg (s.e.m. 0.79). This preserved the previously achieved weight reduction with an average difference of approximately 3 kg. Among all participants in cohort 1 randomized only to orforglipron who achieved 15% or more body weight reduction in SURMOUNT-5, 63.7% (s.e.m. 5.03) maintained 15% or more body weight reduction after 52 weeks of treatment with orforglipron.
Sensitivity analysis of weight change from SURMOUNT-5 (SM-5) to ATTAIN-MAINTAIN for cohort 1 (tirzepatide (TZP) to randomized control trial). a, Cohort 1: percent change in body weight from SURMOUNT-5 to ATTAIN-MAINTAIN. Observed data and efficacy estimand. b, Cohort 1: mean change in body weight from SURMOUNT-5 to ATTAIN-MAINTAIN. Observed data and efficacy estimand. c, Cohort 1: absolute body weight from SURMOUNT-5 to ATTAIN-MAINTAIN. Observed data and efficacy estimand. Data presented in line plots are observed mean (s.e.m.). *Data are MBE (s.e.m.) for the mean actual value of body weight based on MMRM analysis. Dashes beginning at week 24 include participants eligible for rescue therapy. Data in ATTAIN-MAINTAIN after rescue orforglipron were imputed with worst value observed before start of rescue. All other data are MBE (s.e.m.) of the modified intent-to-treat population.
Source data
Participants in cohort 1 randomized only to orforglipron in ATTAIN-MAINTAIN had a mean baseline waist circumference of 120.8 cm (s.e.m. 1.48) in SURMOUNT-5. At the start of the ATTAIN-MAINTAIN study, they had a mean waist circumference of 100.3 cm (s.e.m. 1.50) with a decrease in mean waist circumference of 20.5 cm (s.e.m. 0.95) (Extended Data Fig. 2a,b). Based on the efficacy estimand, participants in cohort 1 had a mean decrease in waist circumference of 16.3 (s.e.m. 0.86) cm from the beginning of SURMOUNT-5, resulting in an MBE waist circumference of 103.4 cm (s.e.m. 0.86) with orforglipron. This preserved their previously achieved decrease in waist circumference with an average change from randomization of approximately 3 cm (Extended Data Fig. 2a).
In addition, other cardiometabolic risk factors demonstrated similar preservation of reductions at the end of ATTAIN-MAINTAIN. As an example, in cohort 1, participants subsequently randomized to orforglipron had a mean baseline HbA1c of 5.6% at the beginning of SURMOUNT-5. At the beginning of ATTAIN-MAINTAIN, after weight reduction with injectables, the HbA1c improved to a mean of 5.2%. At 52 weeks, the mean HbA1c remained at 5.2% and retained the improvements in this marker after switching to oral orforglipron. Similar trends were observed in insulin levels, fasting serum glucose (FSG), markers of lipids such as triglycerides and non-HDL, and systolic blood pressure (Extended Data Fig. 3a–e).
Cohort 2: semaglutide to orforglipron (MTD) versus placebo
For the primary endpoint with modified treatment-regimen estimand, participants in cohort 2 who achieved body weight plateau maintained 79.3% (s.e.m. 4.42) of body weight reduction with orforglipron compared with 37.6% (s.e.m. 7.46) with placebo, with an ETD relative to placebo of 41.7 (95% CI 24.4 to 59.0); P < 0.001) at week 52. Among key secondary endpoints in cohort 2, participants who achieved a body weight plateau, 55.0% (s.e.m. 5.08) of those treated with orforglipron maintained ≥80% of the body weight reduction achieved during SURMOUNT-5, compared with 6.9% (s.e.m. 3.77) with placebo, corresponding to a risk difference of 48.1 (95% CI 35.6 to 60.5; P < 0.001) (efficacy estimand data shown in Extended Data Fig. 1b). All participants in cohort 2 maintained 85.9% (s.e.m. 4.60) of body weight reduction with orforglipron and 40.2% (s.e.m. 7.87) with placebo, with an ETD relative to placebo of 45.6 percentage points (95% CI 28.3 to 63.0; P < 0.001) with orforglipron treatment. All participants in cohort 2 had a mean MBE percent change from SURMOUNT-5 baseline body weight of –14.9% (s.e.m. 0.94) with orforglipron and –7.9% (SE 0.81) with placebo, with an ETD relative to placebo of –7.0 percentage points (95% CI –9.1 to –5.0; P < 0.001).
In additional sensitivity analyses of the efficacy estimand, participants in cohort 2 randomized only to orforglipron in ATTAIN-MAINTAIN had a mean baseline body weight of 113.5 kg (s.e.m. 2.64) in SURMOUNT-5. At the start of the ATTAIN-MAINTAIN study, they had a baseline body weight of 95.0 kg (s.e.m. 2.51) with a reduction of 16.5% (s.e.m. 0.81). Participants in cohort 2 had a mean MBE body weight reduction of 15.1% (s.e.m. 0.67) (Fig. 3a) and absolute body weight reduction of 17.1 kg (Fig. 3b) from the beginning of SURMOUNT-5 to the end of ATTAIN-MAINTAIN. This represents a difference in percent weight change of approximately 1% and an average difference in weight of approximately 1 kg from baseline. For these participants, the mean body weight (MBE) at week 52 was 95.9 kg (s.e.m. 0.76) with orforglipron (Fig. 3c). Participants in cohort 2 at 24 weeks, before rescue therapy eligibility per study protocol, had a mean MBE body weight reduction of 16.3% (s.e.m. 0.56) from the beginning of SURMOUNT-5, with a mean MBE body weight of 94.5 kg (s.e.m. 0.68). This preserved all of the previously achieved weight reduction. Among all participants in cohort 2 randomized only to orforglipron who achieved 15% or more body weight reduction in SURMOUNT-5, 69.5% (s.e.m. 5.79) maintained 15% or more body weight reduction after 52 weeks of treatment with orforglipron.
Sensitivity analysis of weight change from SURMOUNT-5 to ATTAIN-MAINTAIN for cohort 2 (semaglutide (SEMA) to randomized control trial). a, Cohort 2: percent change in body weight from SURMOUNT-5 to ATTAIN-MAINTAIN. Observed data and efficacy estimand. b, Cohort 2: mean change in body weight from SURMOUNT-5 to ATTAIN-MAINTAIN. Observed data and efficacy estimand. c, Cohort 2: absolute body weight from SURMOUNT-5 to ATTAIN-MAINTAIN. Observed data and efficacy estimand. Data presented in line plots are observed mean (s.e.m.). *Data are MBE (s.e.m.) for the mean actual value of body weight based on MMRM analysis. Dashes beginning at week 24 include participants eligible for rescue therapy. Data in ATTAIN-MAINTAIN after rescue orforglipron were imputed with worst value observed before start of rescue. All other data are MBE (s.e.m.) of the modified intent-to-treat population.
Source data
Participants in cohort 2 randomized only to orforglipron in ATTAIN-MAINTAIN had a mean baseline waist circumference of 119.1 cm (s.e.m. 1.77) in SURMOUNT-5. At the start of the ATTAIN-MAINTAIN study, they had a waist circumference of 103.4 (s.e.m. 1.83) cm with a decrease in mean waist circumference of 15.6 (s.e.m. 1.09) cm (Extended Data Fig. 2c,d). Based on the efficacy estimand, participants in cohort 2 had a mean decrease in waist circumference of 14.2 (s.e.m. 0.90) cm from the beginning of SURMOUNT-5, resulting in an MBE waist circumference of 104.5 (s.e.m. 0.90) cm with orforglipron. This preserved their previously achieved decrease in waist circumference with an average change from randomization of approximately 1 cm (Extended Data Fig. 2c).
In addition, other cardiometabolic risk factors demonstrated similar preservation of reductions at the end of ATTAIN-MAINTAIN. As an example, in cohort 2 participants subsequently randomized to orforglipron had a mean baseline A1c of 5.6% at the beginning of SURMOUNT-5. At the beginning of ATTAIN-MAINTAIN, after weight reduction with injectables, the A1c improved to a mean of 5.2%. At 52 weeks, the mean A1c remained at 5.2% and retained the improvements in this marker after switching to oral orforglipron. Similar trends were observed in insulin levels, FSG, markers of lipids such as triglycerides and non-HDL, and systolic blood pressure (Extended Data Fig. 4a–e).
Rescue therapy
Starting at week 24, participants on placebo who regained 50% or more of the weight loss achieved during the SURMOUNT-5 trial were initiated on orforglipron as rescue therapy. Participants began taking rescue orforglipron 1 mg orally each day, with dose escalation every 4 weeks until the MTD was achieved as determined by the interactive web response system. For participants taking orforglipron 24 mg, they were allowed to re-escalate to orforglipron 36 mg using the interactive web response system to maintain blinding. Other oral OMMs were not permitted as rescue therapy in the study. In those participants who had ≥50% weight regain in cohort 1 and cohort 2, 39 (65.0%) and 42 (64.6%) received rescue orforglipron therapy in the placebo group, respectively. In those randomized to the orforglipron group, two participants in cohort 1 and one participant in cohort 2 were escalated from a dose of 24 mg to 36 mg of orforglipron. Furthermore, at 52 weeks, only 25 participants (31.3%) from cohort 1 and 12 participants (18.2%) from cohort 2 who were originally randomized to placebo completed treatment without receiving active therapy (either as rescue or other OMMs).
Safety
The proportion of participants reporting any adverse events (AEs) and the number of reported serious adverse events (SAEs) were similar between arms (Table 2; see additional safety data in Extended Data Tables 1 and 2). Across both the cohorts, study drug discontinuations owing to AEs were 4.8% to 7.3% with orforglipron MTD. The most frequently reported AEs with orforglipron were gastrointestinal disorders of nausea, constipation, vomiting or diarrhea. The overall incidence of GI AEs including nausea, vomiting or diarrhea in the first 4 weeks of the trial for both cohorts was 10.5% and 9.5% in the tirzepatide and semaglutide cohorts, respectively. Most GI AEs were mild to moderate in severity, and there were no dose de-escalations within the first 4 weeks when transitioning from injectable therapy directly to 12 mg of orforglipron (Extended Data Fig. 5).
SAEs were reported by three participants in the orforglipron group in cohort 1 and one participant each in the orforglipron and placebo group in cohort 2, accounting for <2% of the total group (Table 2). One case of mild pancreatitis was confirmed through adjudication in the orforglipron treatment group in cohort 1. Among the participants treated with orforglipron in cohort 1, four participants had increased ALT levels greater than or equal to three times the upper limit of normal (ULN) and one participant in cohort 2 and one participant in the placebo group for both cohorts 1 and 2 experienced this elevation in ALT (Extended Data Table 3). In both cohorts 1 and 2, two participants receiving orforglipron had increased AST levels greater than or equal to three times the ULN. In the placebo group, one participant in cohort 2 experienced this elevation (Extended Data Table 3). Of the participants experiencing these aspartate aminotransferase (AST)/alanine aminotransferase (ALT) elevations, one participant in cohort 2 receiving placebo also experienced an increase of twice the ULN for total bilirubin. Overall, no liver safety signals were detected. One death was reported in the orforglipron group in cohort 2 and was not considered related to the study treatment.