Ethical approval statement
The study protocol, all study protocol amendments, written study participant information, informed consent form, Investigator’s Brochure and any other relevant documents were reviewed and approved by an independent ethics committee or institutional review board at each study site.
This study was conducted in accordance with the study protocol and all approved amendments; the International Council for Harmonisation (ICH) Guideline for Good Clinical Practice E6(R2); the ethical principles of the Declaration of Helsinki; the Council for International Organizations of Medical Sciences, International Ethical Guidelines; all applicable local laws and regulations; and ICH requirements for archiving and retention of essential documents.
Study design and oversight
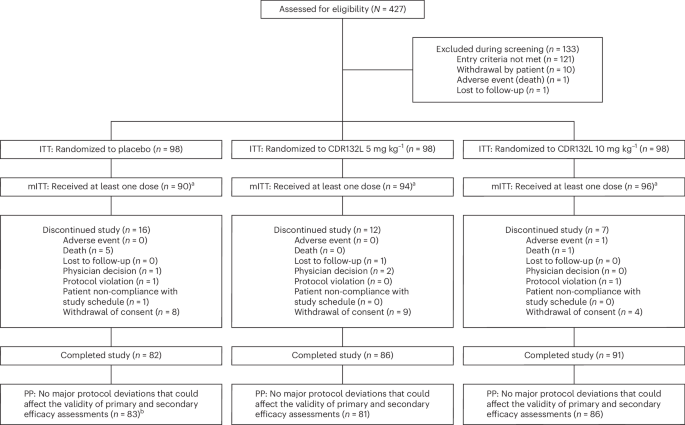
HF-REVERT was an international, multicenter, randomized, double-blind, placebo-controlled, parallel-group phase 2 trial evaluating two dose levels of CDR132L in patients with LV systolic dysfunction after MI. The trial was conducted at 54 study sites (that is, study sites that consented at least one patient) in eight countries (Czech Republic, Germany, Greece, Hungary, The Netherlands, Poland, Spain and the United Kingdom). Recruitment took place between July 2022 and March 2024.
Patients were assigned in a 1:1:1 ratio to CDR132L 5 mg kg−1, CDR132L 10 mg kg−1 or placebo, administered as three intravenous infusions 28 days apart, on top of contemporary standard of care. The trial consisted of a screening period (3–14 days after MI diagnosis), a 6-month double-blind treatment period and a 6-month follow-up, with the end-of-study visit at month 12.
The trial was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines. All patients provided written informed consent. An independent data monitoring committee oversaw patient safety. Echocardiographic imaging was analyzed at a blinded core laboratory at Brigham and Women’s Hospital, Harvard Medical School, using standardized acquisition and interpretation protocols.
The trial protocol and the statistical analysis plan are available in Supplementary Note 1 and Supplementary Note 2, respectively.
Patients
Eligible participants were adults aged 30–80 years with a spontaneous type 1 MI (STEMI or non-ST-segment elevation myocardial infarction (NSTEMI)), randomized 3–14 days after the index event. Patients were required to have an LVEF ≤45% confirmed on core laboratory echocardiography, NT-proBNP ≥125 pg ml−1 and <8,000 pg ml−1 and significantly elevated troponin levels (for NSTEMI, ≥5× the upper limit of normal). All patients underwent percutaneous coronary intervention or diagnostic angiography for the index event.
Major exclusion criteria included non-ischemic cardiomyopathy; prior LVEF <30% within 6 months; New York Heart Association (NYHA) Class IV; planned cardiac surgery; severe valvular disease; hemodynamic instability; significant renal impairment (estimated glomerular filtration rate <30 ml min−1 1.73 m−2) or hepatic impairment (Child–Pugh B/C); active viral infections; recent major neurological events; thrombocytopenia; uncontrolled diabetes; epilepsy; unstable psychiatric or medical conditions; and clinically significant electrocardiogram abnormalities.
The complete inclusion and exclusion criteria are provided in Extended Data Table 4. Data on gender were not collected. Some further exploratory analyses were performed stratified by sex for key endpoints, including LVESVI and LVEF, but the small number of treated women does not allow a meaningful conclusion.
Randomization and masking
Patients were randomized centrally using a computer-generated schedule stratified by age group (<60 years, ≥60 years) and location of infarction (anterior, non-anterior). Investigators, patients, core laboratory personnel and the sponsor remained blinded until all patients had completed month 6. The study drug was administered in a double-blind fashion whereby patients and clinical study site staff members were blinded to the study treatment. The pharmacy staff members who prepared the study drug were not blinded to study drug assignment.
Echocardiographic assessment
Transthoracic echocardiograms were obtained at baseline and at months 3, 6 and 12 using a standardized imaging protocol. All study sonographers at study sites were sufficiently trained and had to be certified before study start. All echocardiograms obtained at study sites were transferred to the Brigham/Harvard core laboratory for blinded analysis.
LVESVI and LVEF were quantified using the Simpson’s rule algorithm.
Other echocardiographic parameters were assessed by the core laboratory’s subset of standard echocardiographic examinations.
miRNA measurements in plasma
Plasma miR-132 levels were quantified at Biotype GmbH using the CardiorHealth miR-132 Plasma PCR Kit (Cardior Pharmaceuticals GmbH) in accordance with the manufacturer’s instructions. The assay is a CE-marked in vitro diagnostic based on quantitative real-time polymerase chain reaction (PCR) for specific detection of miR-132-3p in plasma from patients with established heart failure or at risk of heart failure after MI.
Total RNA was isolated from 150 µl of EDTA plasma spiked with cel-miR-39-3p using the miRNeasy Serum/Plasma Advanced Kit (Qiagen). Reverse transcription was performed in a single reaction using the TaqMan MicroRNA Reverse Transcription Kit (Thermo Fisher Scientific) with miR-132-3p and cel-miR-39-3p specific reverse transcription primers. Quantitative PCR was carried out on a LightCycler 480 II instrument (Roche) using unlabeled forward and reverse primers and a FAM-labeled TaqMan MGB probe, with each microRNA analyzed in separate reactions on the same plate. Results were reported as Cp values, and relative plasma miR-132 levels were calculated as 2 − ΔCp (2 − (Cp miR-132 – Cp cel-miR-39)).
Statistical analysis
The primary endpoint was the percentage change in LVESVI from baseline to month 6. The primary analysis used an analysis of covariance (ANCOVA) model that included treatment as a fixed effect and baseline LVESVI as a covariate. The analysis was also adjusted for center group and stratification factors of age group (<60 years, ≥60 years) and location of infarction (anterior, non-anterior). The LSM estimates for each treatment arm are displayed together with standard errors and their corresponding 95% confidence intervals. Treatment differences with 95% confidence intervals were also produced.
All observed data were included in the analysis regardless of post-randomization events such as treatment discontinuations or hospitalizations for CRT implants or heart transplantations. Missing post-baseline data through month 6 were imputed using the last observation (including baseline) carried forward (LOCF) approach. In addition, MMRM was used for sensitivity analyses. The model used treatment, timepoint, treatment-by-month interaction, center group, stratification factors of age group (<60 years, ≥60 years) and location of infarction (anterior, non-anterior) and baseline LVESVI.
The primary analysis set was the mITT population (all randomized patients who received at least one dose of study drug (CDR132L or placebo)). The safety population comprised all randomized patients who received at least one dose of study drug and had at least one post-dose safety assessment. Prespecified supportive analyses were performed in addition in the PP population, which included patients who completed treatment and 6-month visit without any major protocol deviations.
Supportive analyses were performed for the PP population in a similar manner to the primary efficacy analysis.
Predefined subgroup analyses of the primary endpoint were performed at month 6 and month 12 in the mITT and PP populations. Subgroup analyses of the secondary endpoints (that is, absolute and relative change from baseline in LVESVI, LVEF and NT-proBNP) were also performed at months 6 and 12 in the mITT and PP populations.
Predefined exploratory subgroup analyses based on age group, sex, MI type, time of first treatment after MI event, patients with and without treatment of SGLT2 inhibitors and disease severity, including patients with baseline LVEF values and NT-proBNP concentrations above/below the median, were performed in an exploratory fashion for secondary endpoints.
However, only one important post hoc-defined subgroup was analyzed: patients post-MI with baseline LVESVI above/below the median. This subgroup was analogous to predefined subgroups of LVEF and NT-proBNP (above/below median), and it was analyzed in a similar manner to the primary efficacy analysis. The subgroup analysis used an ANCOVA model that included treatment and center group, stratification factors of age group (<60 years, ≥60 years) and location of infarction (anterior, non-anterior), baseline LVESVI, subgroup and treatment-by-subgroup interaction.
All analyses performed used SAS version 9.4 software.
Sample size determination
Sample size was determined using a one-sided two-sample t-test with a significance level of 2.5%. Based on assumed mean LVESVI changes of 1% for placebo, 5% for CDR132L 5 mg kg−1 and 6% for CDR132L 10 mg kg−1, each with a common s.d. of 9%, a sample size of 90 patients per group provided 96.0% power to detect a 5-percentage-point difference between 10 mg kg−1 and placebo (step 1) and 84.3% power to detect a 4-percentage-point difference between 5 mg kg−1 and placebo (step 2), within a hierarchical testing procedure. This resulted in an overall power of 80.9%. To account for early dropouts, the planned sample size was increased to 294 patients.
Secondary endpoints
Secondary endpoints (changes from baseline to months 6 and 12 in LVEF, NT-proBNP, troponin T, GLS and KCCQ, and change from baseline to month 12 in LVESVI) were analyzed using ANCOVA, with change scores expressed as LSMs with standard errors and 95% confidence intervals. No adjustment for multiple comparisons was prespecified.
Safety
The safety of CDR132L was assessed by continuous monitoring of adverse events and abnormalities in clinical laboratory assessments, vital signs, physical examination, electrocardiograms and urinalysis.
Adverse events were reported by the patient (or, when appropriate, by a caregiver, surrogate or the patient’s legally authorized representative).
The investigator was responsible for detecting, documenting and recording events that met the definition of an adverse event or a serious adverse event and remained responsible for following-up adverse events that were serious, that were considered related to the study treatment or study procedures or that caused the patient to discontinue the study treatment.
Adverse events were summarized with the number of patients, percentage of patients, number of events and the rate of events.
The rate of events was calculated as (number of events / patient years of exposure), where patient years of exposure = (last dose date – first dose date + 31) / 365.25.
At each level of summarization, a patient was counted once if he/she reported one or more events. The severity grade and relationship to study drug were summarized in a similar manner.
All safety analyses were based on the safety population. No formal statistical analysis of the safety data was performed.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.