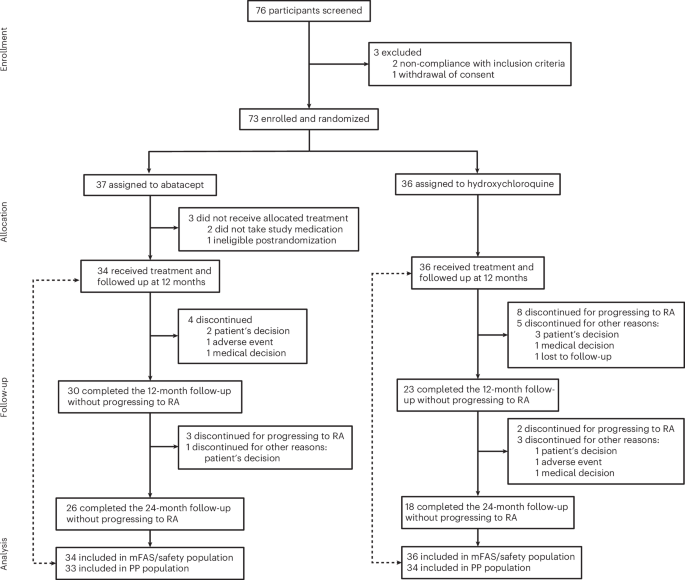
The PALABA trial (effects of abatacept on the progression to RA in patients with palindromic rheumatism) was a randomized, open-label trial conducted at 14 rheumatology centers in Spain. The protocol was approved by the ethics committee of the Hospital Clínic Barcelona (Barcelona, Spain; Reference HCB/2018/0768) and the Spanish Agency of Medicines (Reference 2018 MUH/CLIN/EC). This study was conducted in accordance with the principles of the Declaration of Helsinki and Good Clinical Practices. All participants provided written informed consent before inclusion in the study. The trial was registered at ClinicalTrials.gov (NCT03669367) and EudraCT (no. 2017-004543-20). The study protocol is available as Supplementary Information.
Participants
We included adults aged ≥18 years who were diagnosed with PR according to the modified criteria of Guerne and Weissman34, who had a disease duration of ≥3 months but <3 years and who were positive for RF and/or ACPA. The modification of the criteria for PR consisted of considering at least a 3-month history instead of a 6-month history of recurrent episodes (see Supplementary Box 1 for the original criteria). Individuals were excluded if they had persistent arthritis (involvement in one or more joints lasting for more than 1 week), met the criteria for other rheumatic diseases, had evidence of radiographic damage, had previously used conventional synthetic or biologic DMARDs, were receiving glucocorticoids within 1 month before study entry, had any contraindication to the study drugs, were pregnant or wished to become pregnant, or were not willing to take appropriate contraceptive measures during the study.
Randomization and masking
Randomization codes were assigned centrally using electronic Case Report Forms at the time of patient inclusion. These randomized codes were produced by PROC PLAN of the SAS, with a 1:1 ratio of assignment between both arms, in blocks multiple of two elements and stratified by center. The center was included as a logistic stratification factor in the randomization procedure but was not included as a covariate in the main analysis following regulatory recommendations regarding the use of covariates35.
The participants, clinicians and outcome assessors were not blinded to the treatment assignment.
Procedures
Participants were randomized to abatacept (125 mg subcutaneous injections weekly) or oral hydroxychloroquine (5 mg kg−1 per day) for the first 12 months of treatment; subsequently, abatacept was administered at a dosage of 125 mg every 2 weeks, and hydroxychloroquine was maintained at the same dosage. The reason for maintaining the dose for 2 years was that the progression to RA in individuals with PR is higher during the first 2–3 years36 after the onset of symptoms. Because in the early phase of PR the inflammatory burden is low, following one of the strategies in patients with RA for optimizing biologic therapies once remission is achieved, we decided to reduce the dose of abatacept for the second year of treatment (that is, administration every 2 weeks). Systemic glucocorticoids at an equivalent dosage higher than 7.5 mg per day of prednisone and other DMARDs were not permitted during the study. Patients who met the criteria for RA at any time throughout the study discontinued the protocol and were treated according to the clinician’s criteria.
Participants were seen at screening, baseline and every 3 months thereafter until the end of the study or at the time of treatment discontinuation or early withdrawal. Concomitant medication, general physical examinations, characteristics of the attacks as elicited from the anamnesis (that is, number, duration, intensity as evaluated using a visual analog scale, presence and perceived inflammatory joint signs, and joints involved), joint assessments and assessments of the American College of Rheumatology/European League Against Rheumatism criteria for RA were recorded at each study visit; although it was not explicitly stated in the question, it was understood that ‘intensity’ essentially referred to the degree of pain and/or functional impairment experienced by the patients. Radiographs of the hands and feet were completed at screening, except if they had been performed within the previous 6 months. Blood tests for evaluating blood counts, serum chemistry, C-reactive protein levels and erythrocyte sedimentation rates were performed at screening, at 6 months and every 6 months thereafter. Serum biomarkers were tested at baseline and at 3, 12 and 24 months.
For evaluating the effect of study treatments on AMPAs, we determined by means of appropriate enzyme-linked immunosorbent assay methods seven antigens (Supplementary Methods) with different post-translational modifications: (1) peptides bearing one post-translational modification that included citrullinated peptides derived from vimentin, α-enolase and fibrin and filaggrin (chimeric fibrin and filaggrin citrullinated peptide), and homocitrullinated peptide (chimeric fibrin and filaggrin homocitrullinated peptide); (2) peptides bearing multiple post-translational modifications that included chimeric fibrin and filaggrin citrullinated and homocitrullinated peptide and chimeric fibrin and filaggrin citrullinated and homocitrullinated and acetylated peptide; and (3) the protein antigen carbamylated fetal calf serum. We used a peptide with an unmodified basal structure, chimeric fibrin and filaggrin peptide, as a control peptide. For each antigen specificity, we determined three isotypes: IgG, IgA and IgM. The effects of abatacept and hydroxychloroquine on autoantibody levels, specificities and isotypes were evaluated at baseline and at 3, 12 and 24 months of follow-up for each patient included in the study.
Outcomes
The primary outcome was the development of persistent arthritis (that is, lasting more than 1 week in the same joint) that fulfilled the 2010 RA classification criteria of the American College of Rheumatology/European League Against Rheumatism as evaluated by the participant clinicians during the 24 months of follow-up37.
Secondary outcomes included the frequency, intensity and duration of joint attacks and the proportion of patients in remission, defined as no or a single palindromic attack during at least four consecutive visits (that is, 12 months). We also evaluated the frequency of adverse events. As an exploratory outcome, we evaluated the effect of treatment on AMPA titers.
Statistical analysis
The sample size was calculated to test for progression to RA at any time over 24 months, and the two treatment arms were compared using the chi-square test. Assuming a 2-year progression rate of at least 42% in the control group36 and 11% in the experimental group, a sample size of 35 patients per group (total 70) was necessary to reach a power of 80% at a nominal two-sided alpha level of 0.05, according to the nQuery validated software (nQuery version 9)38. No multiplicity adjustment was implemented. There is only one variable and timepoint defined as a primary endpoint; thus, there is only one confirmatory P value in the trial, and the rest of the P values are considered descriptive at their nominal value.
Primary efficacy analysis was performed using the mFAS, defined as all randomized patients who received at least one dose of the study medication. For the primary outcome, a PP analysis was also performed, which included patients from the mFAS population without major protocol deviations that might impact the main study assessments. Safety analyses were performed in the mFAS population. Missing data on progression to RA were imputed as treatment failure (failure imputation) for primary analysis, and sensitivity analysis was performed using the ADO approach.
Categorical variables are described with absolute and relative frequencies, whereas quantitative variables are described with the mean (s.d.) or the median (P25, P75) if not normally distributed.
For the primary outcome, we conducted a chi-square or Fisher’s exact test, depending on the test requirements. As a supportive analysis, the time to occurrence of RA was estimated using the Kaplan‒Meier method, comparisons were performed with the log-rank test, and HRs (95% CI) were derived from a Cox model. Data were censored at the time of the event, the last documented visit or the end of the follow-up period, whichever occurred first.
For secondary efficacy outcomes, the following strategies were used: the chi-square test or Fisher’s exact test for categorical variables, the dependent or independent t-test for continuous Gaussian-distributed variables and the Mann‒Whitney U-test for ordinal and non-Gaussian continuous data.
Continuous safety variables were analyzed using mixed models for repeated measurements, including a baseline value as a covariate, treatment effect and treatment by visit interaction. The variance‒covariance matrix employed was unstructured. The treatment effect was estimated using adjusted means (least squares mean), s.e.m. and 95% CI. Differences between treatments were estimated on the basis of the differences in the least squares mean, s.e.m. and 95% CI. The participants with at least one adverse event, globally, and by System Organ Class were compared using Fisher’s exact test.
For the analysis of the effect on AMPAs, each antigen–antibody pair was represented as a continuous variable, the Δ optical density. The Δ optical density was calculated by subtracting the optical density of each antigen–antibody pair without post-translational modifications from the corresponding antigen–antibody pair with post-translational modifications. Δ optical density measurements for each antigen–antibody pair at 0, 3, 12 and 24 months were analyzed using a parametric approach (see details below) on previously log-transformed data (GM). Between-group and within-group comparisons were evaluated using the GM ratio and geometric mean fold rise. The parametric approach consisted of a restricted maximum likelihood-based repeated measures approach (mixed models for repeated measurements). Analyses included the fixed, categorical effects for treatment, visit and the treatment-by-visit interactions. A common unstructured (co)variance structure was used to model within-patient errors. If the initial model failed to converge, the following correlation structures were tested in subsequent order until model convergence was achieved: autoregressive model of order 1 (AR[1]), Toeplitz or compound symmetry. The Kenward‒Roger approximation was used to estimate the denominator degrees of freedom39. GM, GM ratio and geometric mean fold rise estimates for each treatment are presented with the associated two-sided 95% CI.
We did not prespecify any subgroup analyses, including sex or gender. To our knowledge, no data suggest that clinical presentation of PR or treatment outcomes differ according to sex or gender. Therefore, sex and gender were not considered in the study design.
Statistical analysis was performed using SAS (version 9.4; SAS Institute Inc.). All the statistical tests were two-sided and were assessed at a significance level of 0.05.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.